
Many of the charges for ions formed by the main group elements can be predicted using the periodic table. The one is usually omitted for ions with a positive or negative one charge.Ītoms will lose or gain specific numbers of electrons when forming an ion. The chemical symbol for fluoride is written as F −. Because it now has one more negative charge than positive charges, it has an overall charge of negative one. Fluorine gains one electron to become a fluoride ion with 9 protons and 10 electrons. Anions are named by adding the suffix '-ide' to their element name.Ī fluorine atom has 9 protons and 9 electrons. Anions have a negative charge because they have more electrons than protons. When atoms or molecules gain electrons, they are called anions. 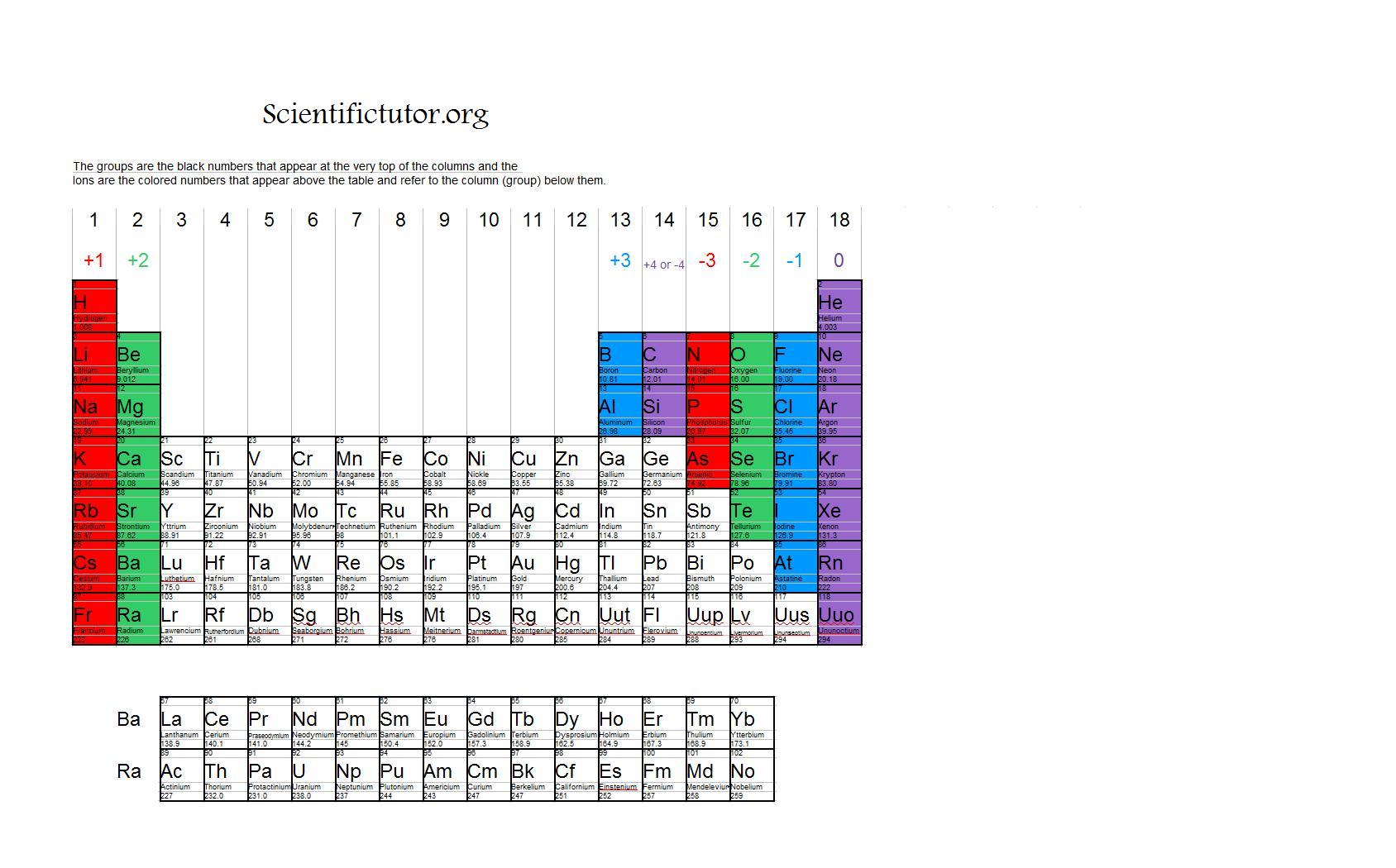
The chemical symbol for a calcium ion is therefore written as Ca 2+. The chemical symbol for an ion is written as the chemical symbol of the atom with the charge written on the right superscript position. Because it now has two more positive charges than negative charges, it has an overall charge of positive two. It loses two electrons to become a calcium ion with 20 protons and 18 electrons. Cations are given the same name as their element, followed by the word ion.Ī calcium atom has 20 protons and 20 electrons. Cations have a positive charge because they have more protons, which have a positive charge, than electrons, which have a negative charge. When atoms or molecules lose electrons, they are called cations. Therefore, the atomic mass of an ion is essentially the same as the neutral atom’s atomic mass. 
Electrons, being much smaller in mass than protons, do not contribute significantly to the atomic mass. When an atom or molecule gains or loses electrons, it is called an ion. An element is defined by the number of protons, which does not change however, many atoms can gain or lose one or more electrons to become a charged particle. 
Atoms are neutral particles with equal numbers of protons and electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
